Real-World Effectiveness of Nirsevimab: Evidence from the NIRSE-GAL Study
Study Design and Methodology of the NIRSE-GAL Study in Galicia
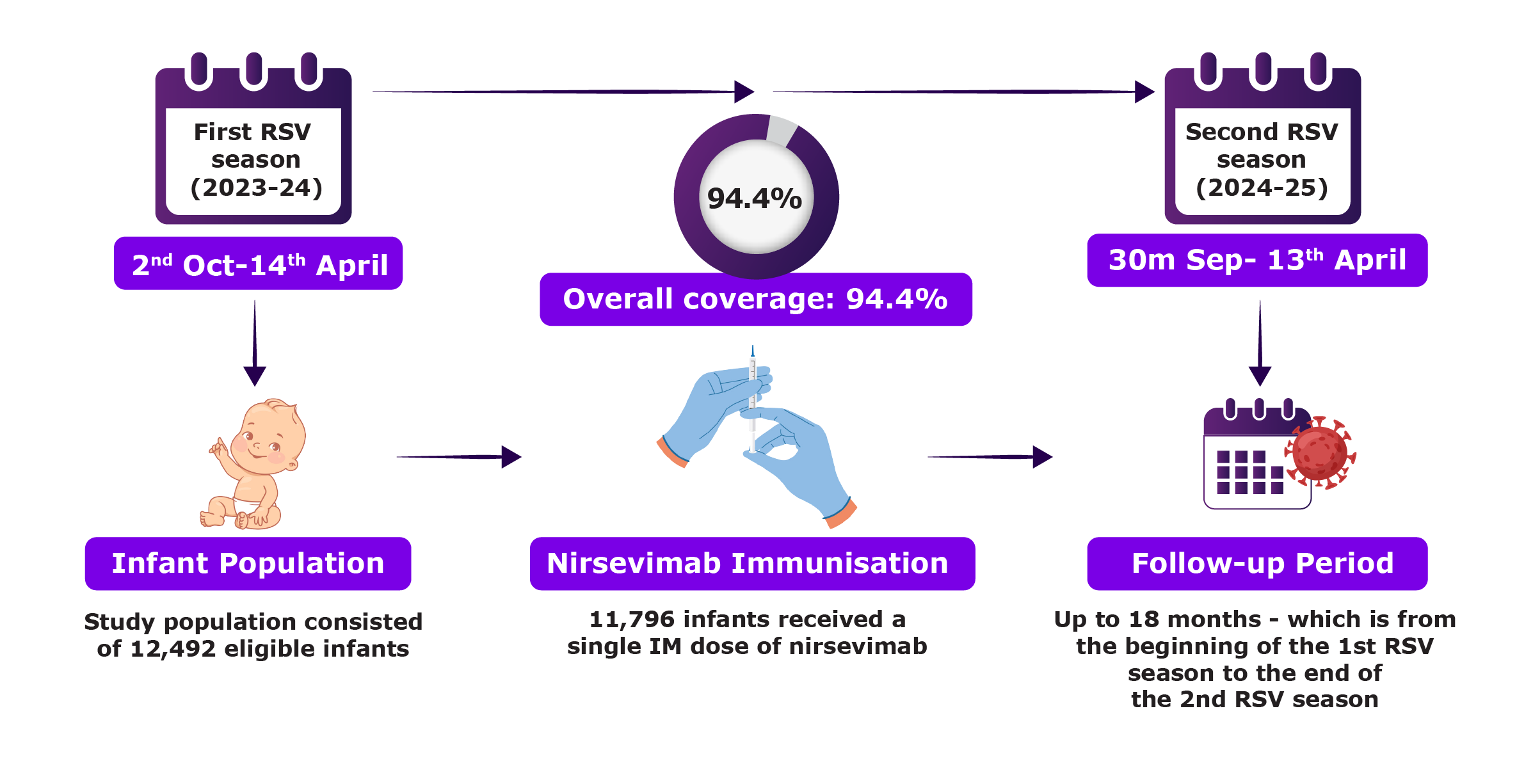
The NIRSE-GAL study is an ongoing population-based, prospective, longitudinal study conducted in Galicia, Spain. It includes infants eligible to receive nirsevimab and evaluates its impact on RSV-related outcomes in both inpatient and outpatient healthcare settings.1
NIRSE-GAL: Population-Based Prospective Study of Nirsevimab in Infants1
IM-Intra-muscular; LRTI-Lower Respiratory Tract Infections; NIRSE-GAL- Nirsevimab Galicia; RSV- Respiratory Syncytial Virus.
Adepted from "Razzini JL et al., 2026"
Clinical Outcomes from the NIRSE-GAL Study in Infants Receiving Nirsevimab
Clinical outcomes were compared with historical RSV seasons before the introduction of nirsevimab to evaluate reductions in RSV-related clinical events. The primary endpoint was RSV-related LRTI hospitalisation.1
Primary Outcome of the NIRSE-GAL Study1
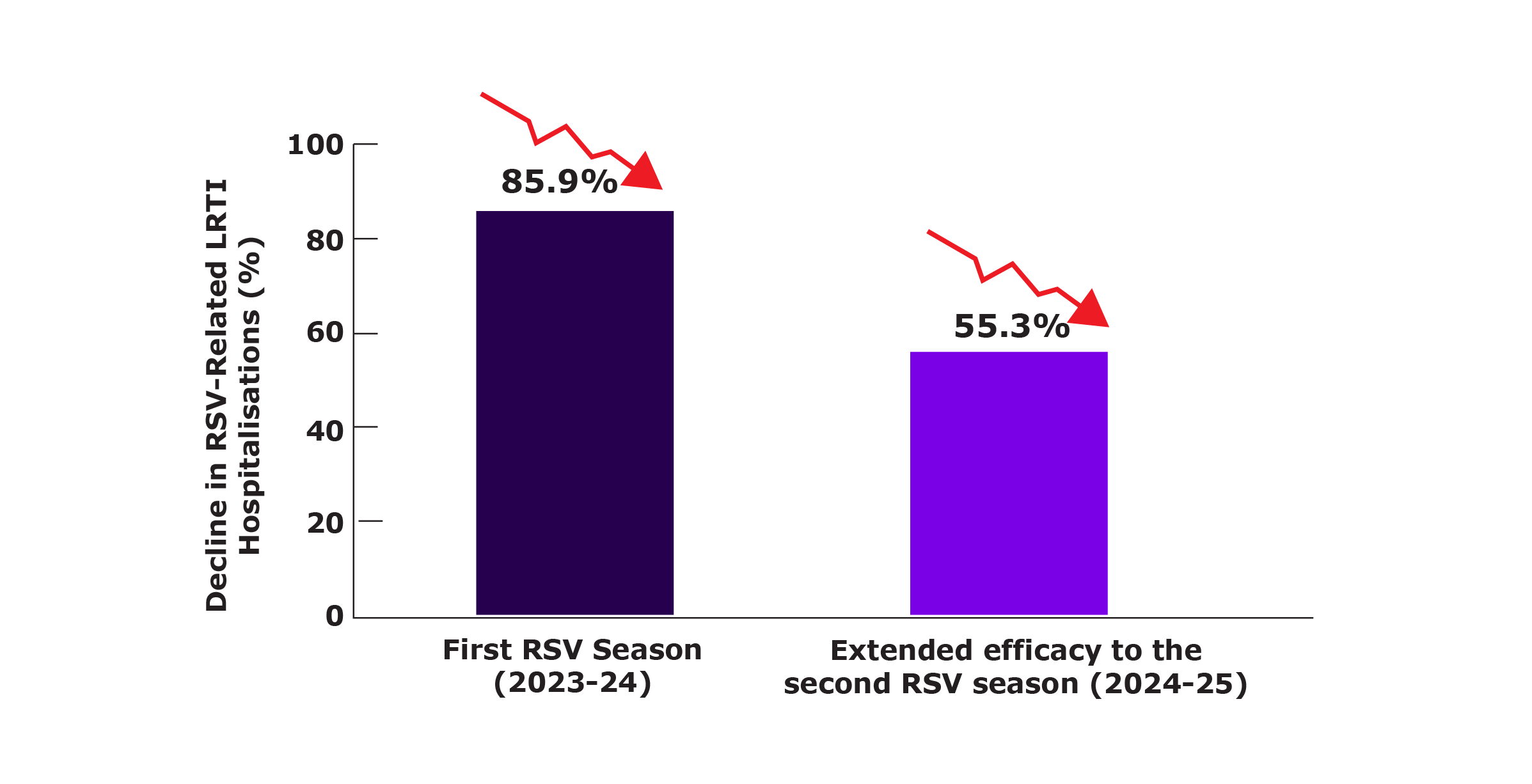
RSV-related LRTI hospitalisations were assessed from their first RSV (2023-24) until the end of their second RSV season (2024-25).
RSV-Related LRTI Hospitalisations Across Two Seasons
Nirsevimab prophylaxis showed sustained reductions in RSV-related LRTI hospitalisations across two seasons.
aLRTI-Lower Respiratory Tract Infections; NIRSE-GAL- Nirsevimab Galicia; RSV- Respiratory Syncytial Virus.
Adapted from "Razzini JL et al.. 2026"
Secondary Outcomes of the NIRSE-GAL Study
Secondary endpoints included LRTI hospitalisation, acute bronchitis or bronchiolitis hospitalisation, and all-cause hospitalisation.1
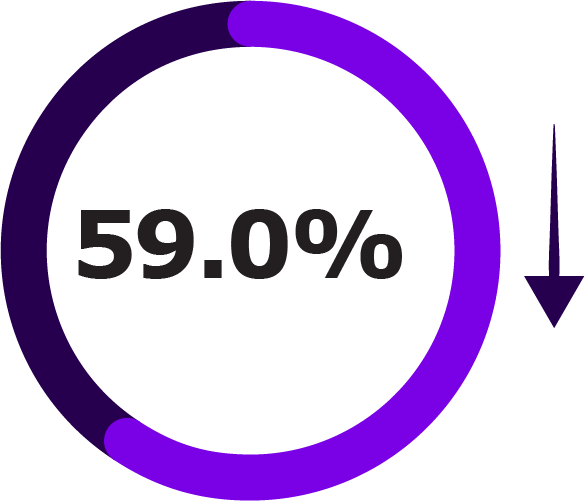
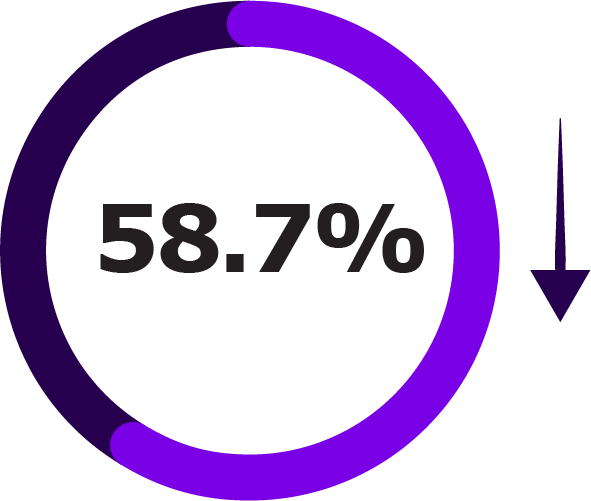
Secondary Outcomes of NIRSE-GAL Study Across Two Seasons1
| First RSV Season Reduction | Up to 18 Months Reduction | |
|---|---|---|
| LRTI hospitalisations | 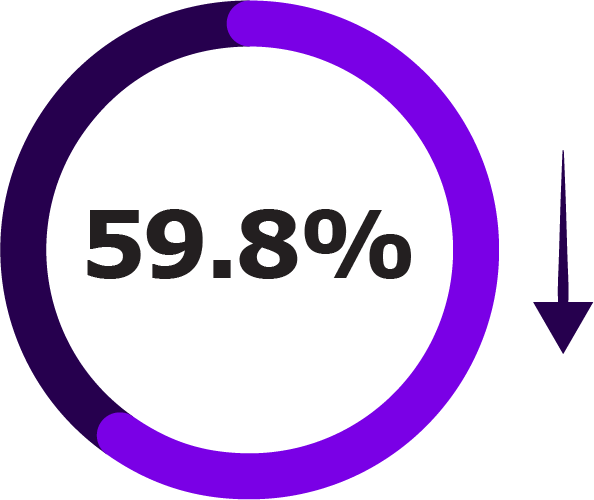
|
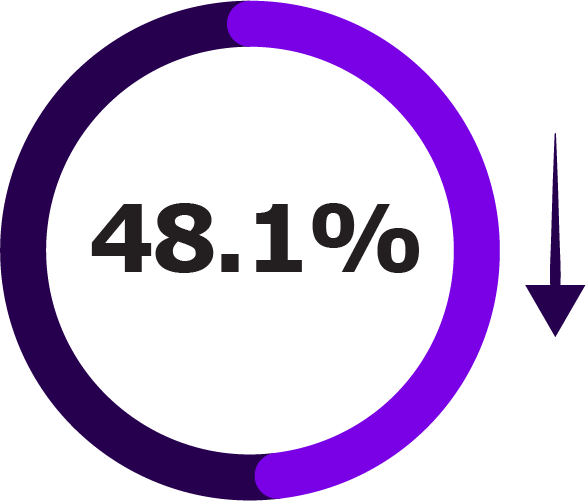
|
| Acute bronchitis or bronchiolitis hospitalisations |
|
|
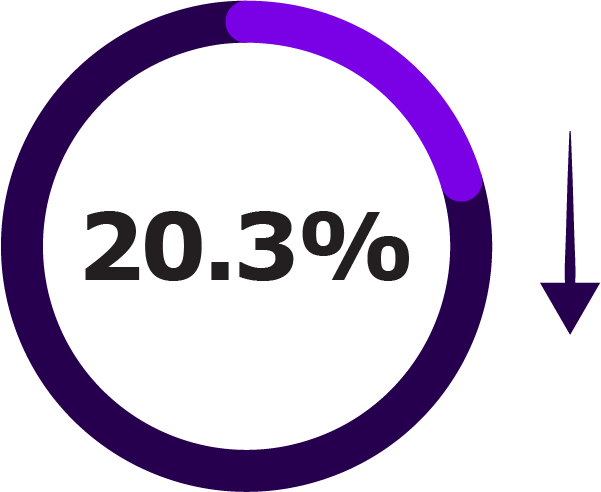
A decrease in all-cause hospitalisations in the first season, indicated a population-level impact beyond RSV-specific outcomes.
LRTI- Lower Respiratory Tract Infections; NIRSE-GAL- Nirsevimab Galicia; RSV- Respiratory Syncytial Virus.
Adapted from "Razzini JL et al., 2026
Recurrence and Outpatient Outcomes
First recurrent hospitalisations in the second RSV season decreased significantly: 78.2% for RSV-related LRTI, 62.4% for LRTI, and 76.9% for acute bronchitis or bronchiolitis.1
Outpatient visits related to respiratory illnesses, including bronchitis, bronchiolitis, wheezing, and asthma, also declined, suggesting a broader reduction in healthcare burden and potential economic benefits.1
Conclusion
Universal nirsevimab prophylaxis in the NIRSE-GAL study significantly reduced RSV-related hospitalisations and outpatient morbidity, with sustained reductions extending into the second RSV season.1 These findings provide strong population-level evidence supporting infant immunization strategies and cost-effectiveness evaluations.1
- Razzini JL, Giné-Vázquez I, Jin J, Santiago-Pérez MI, Pérez-Martinez O et al. Impact of universal nirsevimab prophylaxis in infants on hospital and primary care outcomes across two respiratory syncytial virus seasons in Galicia, Spain (NIRSE-GAL): a population-based prospective observational study. Lancet Infect Dis. 2026 Jan 12:S1473-3099(25)00742-X. doi: 10.1016/S1473-3099(25)00742-X.