{
event: "article_read",
name: `Multiple Myeloma: Minimal Residual Disease`,
author: ``,
tags: ``,
publication_date: ``,
interaction_type: "content"
}
Multiple Myeloma: Minimal Residual Disease
Patients with multiple myeloma who achieve minimal residual disease (MRD) negativity have been shown to have better outcomes compared with MRD positive patients1
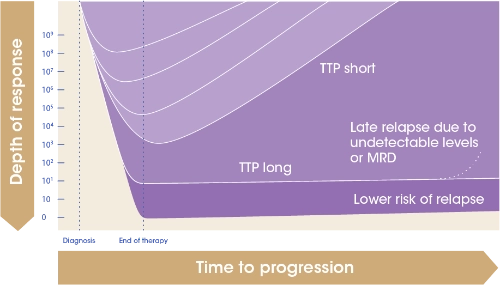
Achieving deeper responses can improve outcomes2
Sustained MRD– = MRD– for 1 year
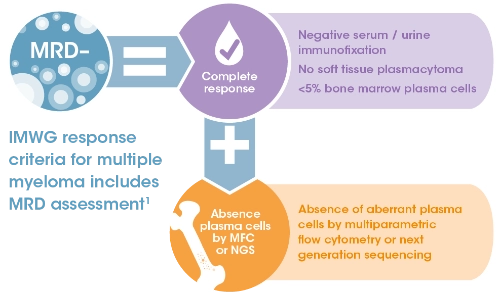
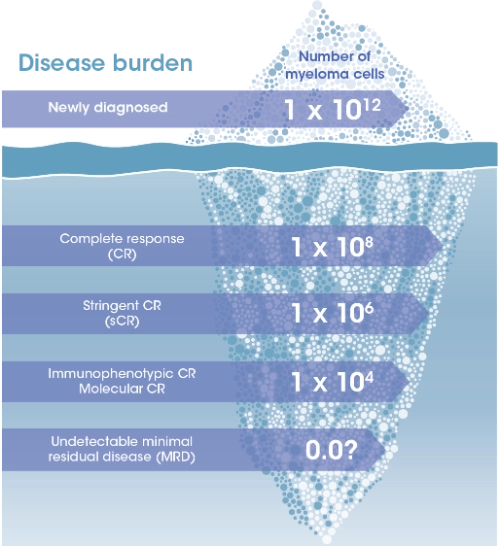
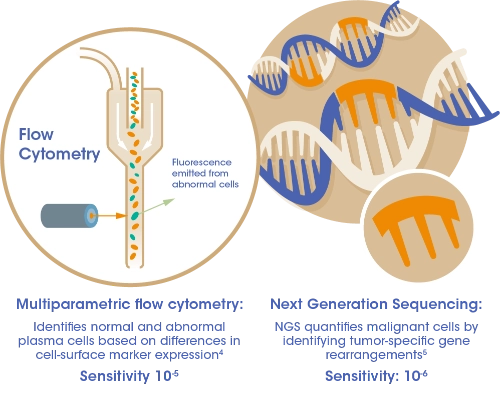
MRD is the low level of malignant cells that persist even after a complete response with myeloma treatment1
The methods used for determining MRD negativity include:
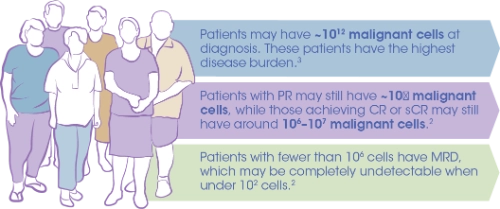
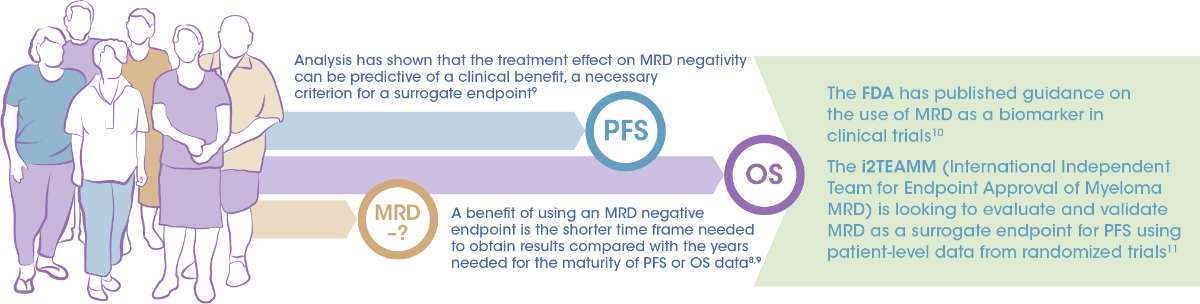
Achieving MRD negativity is associated with better outcomes such as longer PFS and OS compared with patients who are MRD positive6,7
| Meta-analysis of 43 studies involving 8,114 patients: | Meta-analysis of 22 studies involving 4,462 patients: |
MRD negativity is being considered as a regulatory surrogate endpoint for PFS8,9
IMWG, International Myeloma Working Group; NGS, next-generation sequencing; OS, overall survival; PFS, progression-free survival.
- Kumar SK, et al. Lancet Oncol 2016;17:e328–46;
- Paiva B, et al. Blood 2015;125:3059–68;
- Durie BGM, et al. Cancer 1975;36:842–54;
- Flores-Montero J, et al. Leukemia 2017;31:2094–103;
- Ho C, et al. Semin Hematol 2018;55:13–8;
- Munshi NC, et al. JAMA Oncol 2017;3:28–35;
- Munshi NC, et al. Presented at ASH 2019; Poster #4742;
- Avet-Loiseau H, et al. Clin Lymphoma Myeloma Leuk 2020;20: e30–7;
- Landgren O. Semin Hematol 2018;55:1–3;
- Food and Drug Administration. Available at: https://www.fda.gov/media/134605/download;
- Holstein SA, et al. Biol Blood Marrow Transplant 2020;26:e7–15
Related articles
MAT-AE-2300647-V1-Nov-23

.webp/jcr:content/oncology-ta-banner%20(1).webp)
.webp)
.webp)
.webp)
.webp)
.webp)
.webp/jcr:content/Asset%208%20(1).webp)
.jpg/jcr:content/jcr_content%20(14).jpg)

-(1).png/jcr:content/mob-Science%201%20(1)%20(1).png)