BIOCHEMICAL DIAGNOSTIC STRATEGY IN FEMALES SUSPECTED OF FABRY DISEASE
.webp1/jcr:content/science%20hero%20(6).webp)
Study objective and method
To demonstrate
 | The benefits of adding lyso-GL-3 | to ► |  | Primary diagnostic for testing | for ► |
| Improving detection rate of potential females with FD
|
First large prospective study
 | 11,948 females with suspicion of FD tested for |  | Biochemical tests
| followed by |  | Genetic confirmatory testing in 883 females |  |
Groups | 01. Low α-Gal A activity High lyso-GL-3 | 02. Low α-Gal A activity Normal lyso-GL-3 | 03. Normal α-Gal A activity High lyso-GL-3 | 04. Normal α-Gal A activity Normal lyso-GL-3 |
| Classical FD very likely (n=61) | Classical FD unlikely (n=184) | FD very likely (n=256) |  had normal results (n=11,447*) | |
| Genetic confirmatory test | No further testing required | |||
Results
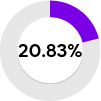 | (184/883) females were identified with one or two GLA variants. |
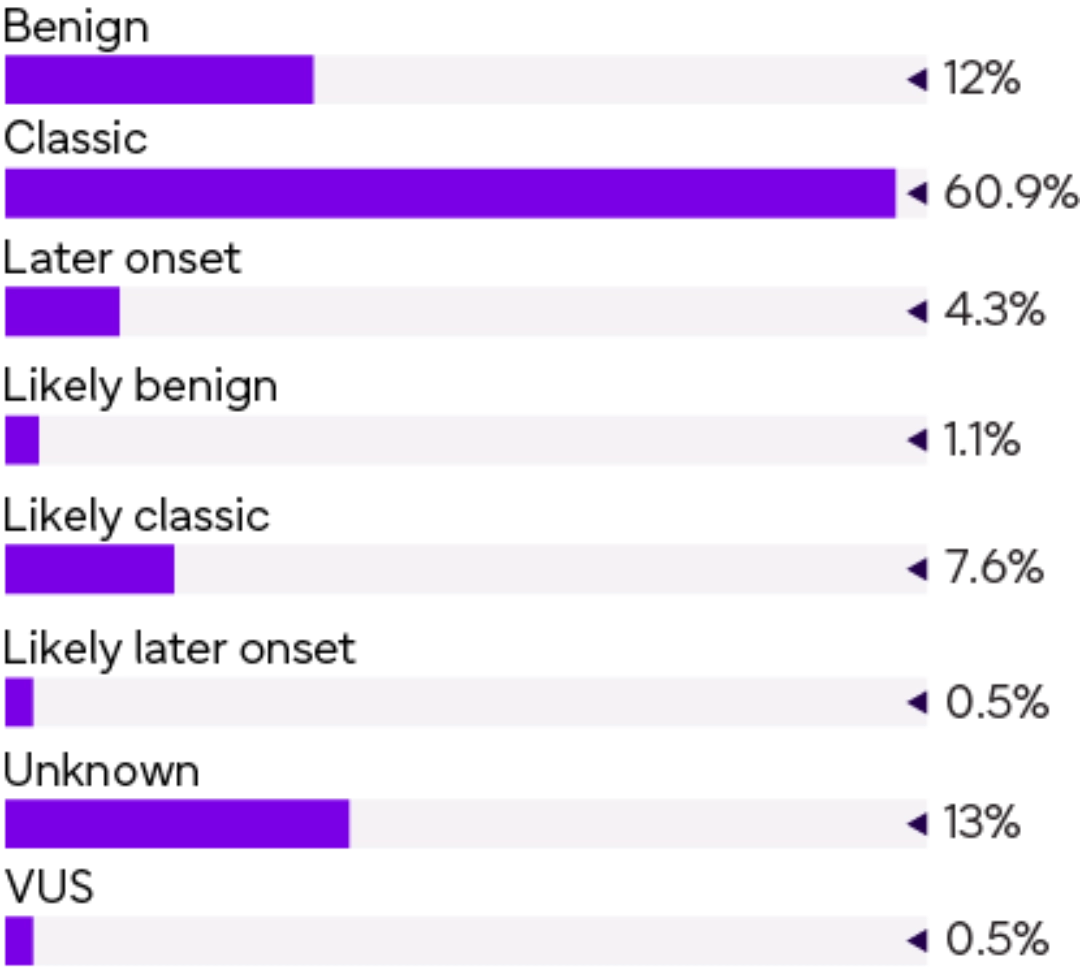
Distribution of genetically positive samples

| Better indicator of FD | |||||
|
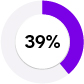
Elevated lyso-GL-3 levels (39% PPV) | > |
Low α-Gal A activity (6% PPV) | Clearly negative results for both biochemical parameters: Unlikely to have FD, even in clinically highly suspicious cases | ||
Proposed diagnosis decision tree

STEP 2
Once enzyme deficiency is confirmed, molecular testing is performed to identify GLA genetic variants.

STEP 2
Once enzyme activity (deficient or not) together with elevated biomarker is confirmed, molecular testing is performed to identify GLA genetic variants.
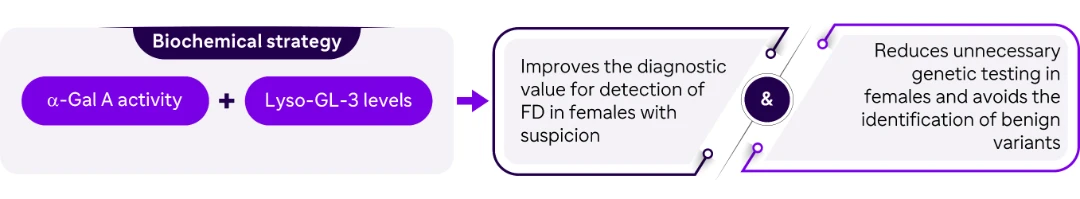
Conclusion
*Mutation analysis performed in 389 cases.
α-Gal A: Alpha-galactosidase A; dbFGP: Fabry disease genotype–phenotype database; FD: Fabry disease; Lyso-GL-3: Globotriaosylsphingosine; PPV: Positive predictive value; DBS: Dried blood spotting; GLA: Galactosidase alpha; VUS: Variant of unknown significance.