Odyssey outcomes - CABG results
Odyssey Outcomes: Addition of PCSK9i to Background Statin Therapy Further Reduces MACE.
CABG Sub-analysis overview
Study Design
- Prespecified analysis
- Patients were categorized by coronary artery bypass graft (CABG) status:
- No CABG (n=16,896)
- CABG after the index ACS but before randomization (index CABG; n=1,025, including n=44 with prior CABG), or
- CABG prior to the index ACS (prior CABG; n=1 ,003)
- The primary composite endpoint of MACE encompassed CHD death, nonfatal MI, ischemic stroke, or unstable angina requiring hospitalization
- The secondary endpoint was all-cause death
Baseline demographics and index events
|
No CABG (n=16,896) |
Index CABG (n=1,025) |
Prior CABG (n=1,003) |
P-value |
|
|
Age, years, median (Q1, Q3) |
58 (51, 65) |
60 (53, 65) |
63 (57, 70) |
<0.0001 |
|
Female, % (n) |
25.8 (4,354) |
20.6 (211) |
19.6 (197) |
<0.0001 |
|
Body mass index, kg/m2, median (Q1, Q3) |
28 (25, 31) |
28 (25, 31) |
29 (26, 32) |
<0.0001 |
|
Region, % (n) |
|
|
|
<0.0001 |
|
Western Europe |
22.3 (3,767) |
22.4 (230) |
17.7 (178) |
|
|
Eastern Europe |
29.9 (5,049) |
17.6 (180) |
20.7 (208) |
|
|
North America |
13.5 (2,273) |
24.1 (247) |
35.0 (351) |
|
|
South America |
13.6 (2,293) |
17.6 (180) |
11.5 (115) |
|
|
Asia |
13.0 (2,196) |
5.5 (56) |
4.1 (41) |
|
|
Rest of the world |
7.8 (1,318) |
12.9 (132) |
11.0 (110) |
|
|
Index event, % (n) |
|
|
|
<0.0001 |
|
NSTEMI |
46.9 (7,913) |
59.8 (612) |
64.9 (650) |
|
|
STEMI |
36.5 (6,156) |
20.9 (214) |
16.6 (166) |
|
|
Unstable angina |
16.6 (2,798) |
19.3 (198) |
18.6 (186) |
|
|
Time from index event to randomization, months, median (Q1, Q3) |
2.6 (1.7, 4.2) |
3.8 (2.7, 6.8) |
2.5 (1.7, 4.1) |
<0.0001 |
NSTEMI, Non-ST Segment Elevation Myocardial Infarction; STEMI, ST Segment Elevation Myocardial Infarction
Lipid-Lowering Therapy at Randomization and Baseline Laboratory Parameters
|
No CABG (n=16,896) |
Index CABG (n=1,025) |
Prior CABG (n=1,003) |
P-value |
|
|
Lipid-lowering therapy at randomization, % (n) |
|
|
|
<0.0001 |
|
High-dose atorvastatin or rosuvastatin |
89.2 (15,077) |
87.8 (900) |
83.2 (834) |
|
|
Other lipid-lowering therapy3 |
10.0 (1,690) |
10.4 (107) |
13.8 (138) |
|
|
No lipid-lowering therapy |
0.8 (129) |
1.8 (18) |
3.1 (31) |
|
|
Lipid measurements, mg/dL, median (Q1, Q3), unless stated |
|
|
|
|
|
LDL-C |
86 (73, 103) |
87 (74, 107) |
94 (78, 116) |
<0.0001 |
|
LDL-C≥100 mg/dL,% (n) |
28.9 (4,878) |
32.5 (333) |
41.7 (418) |
<0.0001 |
|
HDL-C |
42 (36, 50) |
42 (36, 50) |
42 (35, 50) |
0.0166 |
|
Non-HDL-C |
114 (99, 136) |
117 (102, 140) |
126 (107, 153) |
<0.0001 |
|
Triglicerides |
128 (94, 181) |
137 (98, 192) |
139 (102, 195) |
<0.0001 |
|
Apoliproprotein B |
79 (69, 92) |
82 (70, 96) |
85 (75, 103) |
<0.0001 |
|
Lipoprotein(a) |
20.3 (6.6, 58.1) |
27.8 (7.5, 69.0) |
33.2 (8.1, 79.9) |
<0.0001 |
|
Estimated GFR <60 mL/min per 1.73 m2, % (n) |
12.7 (2138) |
13.8 (141) |
25.9 (260) |
<0.0001 |
ᵃ low-/moderate-dose atorvastatin/rosuvastatin, other statin, any lipid-lowering therapy other than statin.
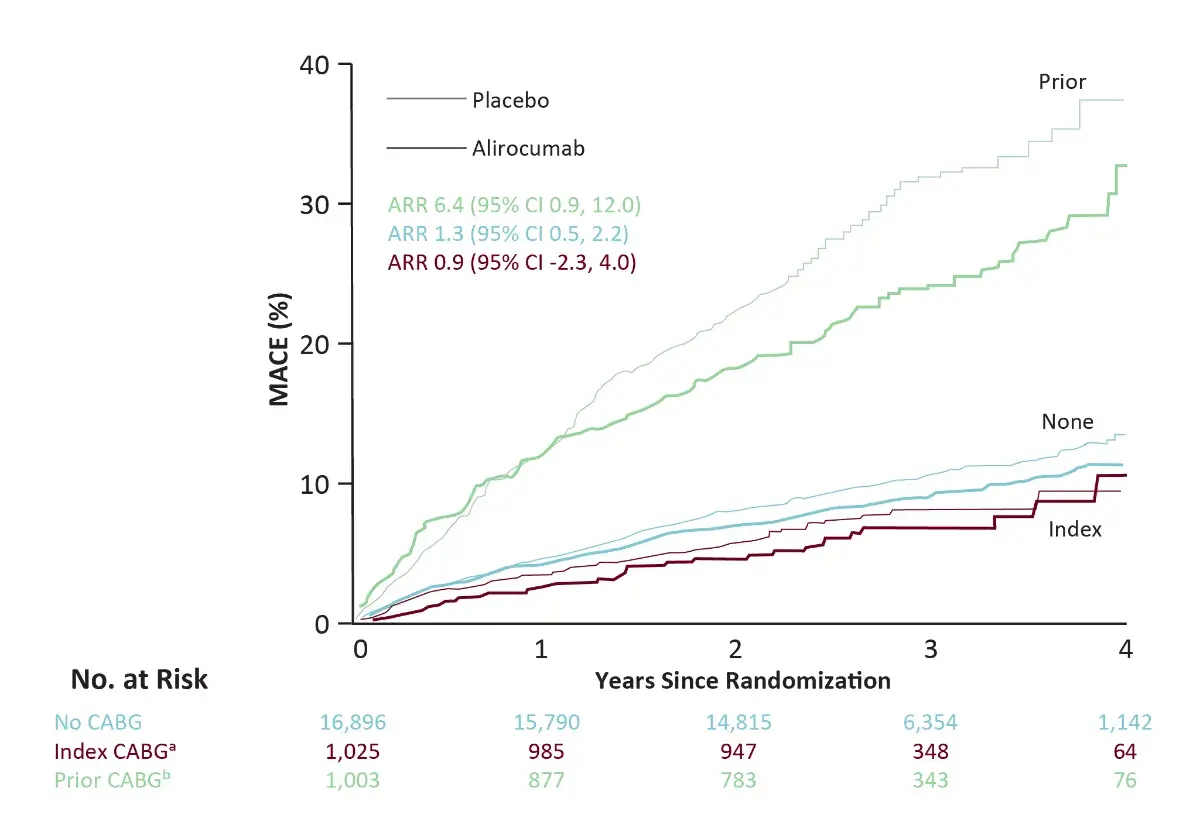
Effect of Alirocumab in patients with ACS, Elevated Atherogenic Lipoproteins and Prior CABG
ᵃ index CABG is CABG between the index ACS event and randomization (including 44 patients with prior CABG);
ᵇ Prior CABG is CABG prior to the index ACS event. HRs reflect stratification by geographic region
CABG Sub-analysis Conclusions
- In this sub-analysis, it was observed that, in a population that has not been fully investigated with regards to the effect of LDL-C lowering therapies, the addition of alirocumab to patients with prior CABG, recent ACS and elevated atherogenic lipoproteins despite intensive statin therapy was associated with consistent relative but greater ARR in MACE and death when compared to patients with an index or no CABG
- Goodman SG, et al. JACC. 2019; 74(9):1177-86.



.webp/jcr:content/ROSETTE%20summary%20(1).webp)
.webp/jcr:content/The%20explorer%20study%20(1).webp)